Comprehensive Pathology Services
End-to-end annotation solutions tailored specifically to the complexity required by pharmaceutical and AI domains.
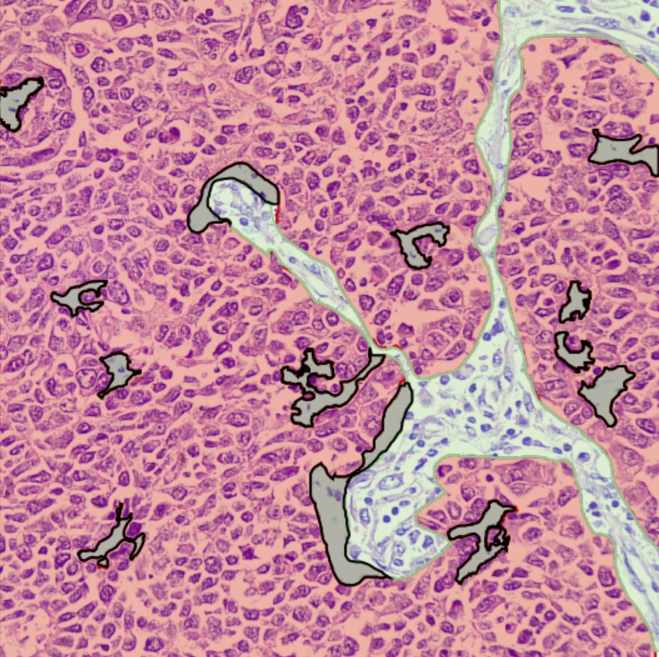
WSI & ROI Segmentation
Region-of-Interest marking, Tumor/Stroma segmentation, Lesion identification, and histological feature segmentation for model training.
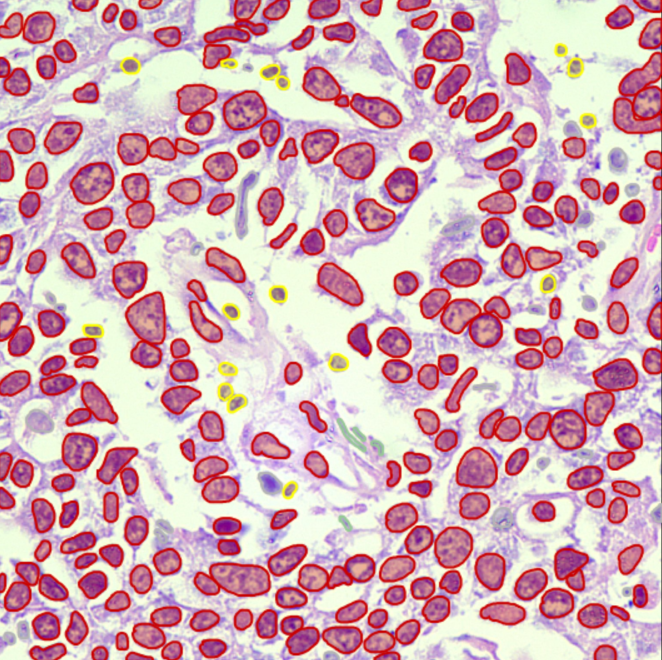
Cellular & Structure Level
High-precision single-cell annotation, mitotic figures, nuclear/cytoplasmic contours, and structure-level tagging.
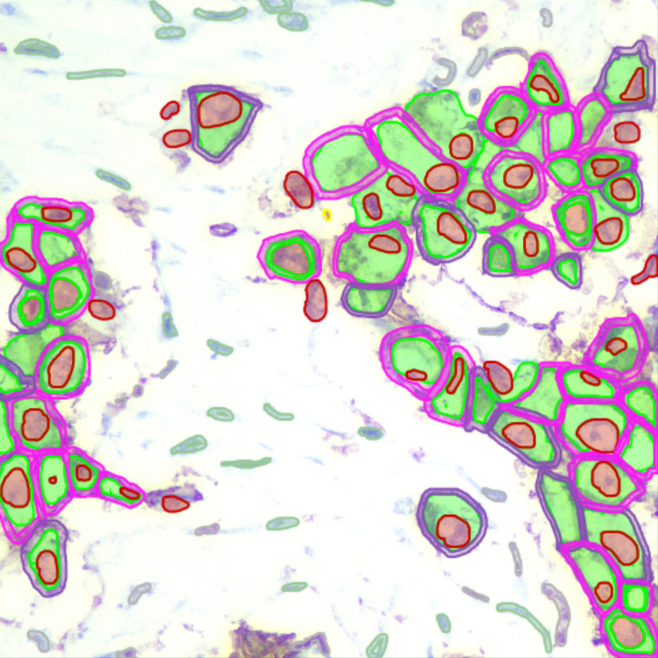
Biomarker Analysis
IHC scoring (Ki-67, PD-L1), ISH/FISH spot counting, and H-Score calculation for drug efficacy studies.
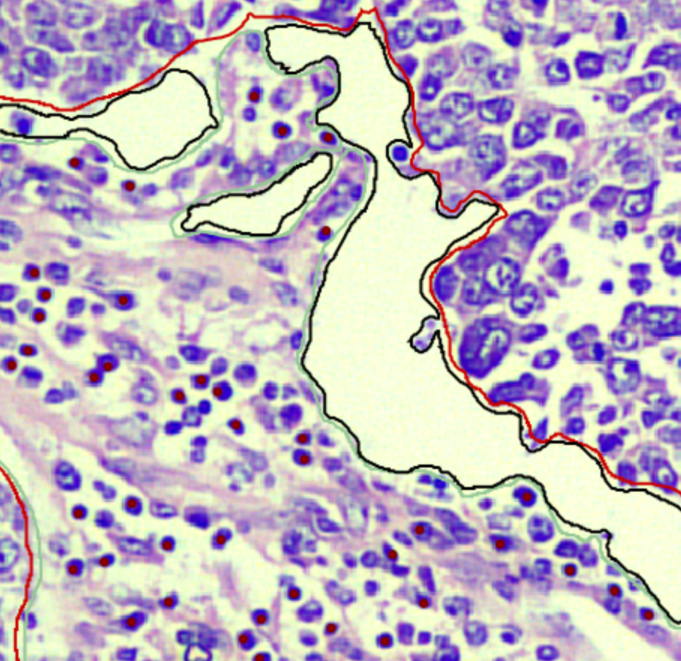
Grading & TME
Lesion grading (Gleason, Nottingham) and Tumor Microenvironment component annotation (TILs, Fibrosis).